Sunday, July 30, 2006
Organometallics, 25 (9), 2248 -2252, 2006
Holly C. Canuto, Admir Masic, Nicholas H. Rees, Stephen J. Heyes, Roberto Gobetto, and Silvio Aime
Abstract:
The novel inclusion compound of -cyclodextrin with the binuclear metal carbonyl complex (5-C5H5)2Fe2(CO)4 as guest molecule is reported. 13C CP/MAS NMR spectroscopy, in the temperature range 100 to 353 K, is used to probe the structure and dynamics of the included molecules. Specifically, below ca. 240 K evidence is presented for the existence of both cis and trans isomers of included (5-C5H5)2Fe2(CO)4. Analysis of the temperature-dependence of the NMR line shapes shows that the microenvironment provided by the -cyclodextrin cavity allows much more extensive dynamic rearrangements of the guest molecules, in comparison to pure cis- or trans-(5-C5H5)2Fe2(CO)4, for which no isomerization or bridging-terminal carbonyl exchange processes are observed in this temperature regime. Notably, even at 100 K, bridging-terminal carbonyl exchange for the included trans isomer is rapid on the exchange-broadening time scale. However, the inclusion cavity is still more dynamically restrictive than a solution environment, and the rates of various exchange processes are usefully modified compared to those detected in solution. For (5-C5H5)2Fe2(CO)4 included in -cyclodextrin, contrary to the situation found in solution, the rate of bridging-terminal carbonyl exchange in the cis isomer is greater than the rate of cis-trans isomerization; in solution direct bridging-terminal exchange in the cis isomer could not be studied because indirect exchange via isomerization to the trans form, which undergoes rapid bridging-terminal exchange, is always significantly faster. By restricting isomerization, the inclusion environment thus confirms for the first time that the cis isomer is capable of carbonyl exchange and would allow the study of its rate and activation parameters.
Organometallics, 25 (14), 3428 -3434, 2006
Convergent Synthesis and Characterization of Organotin Dendrimers Sn{(CH2)nSn[(CH2)4SnPh3]3}4 (n = 3, 4)
Abstract:
The reaction of the haloalkyltin trihalides Br(CH2)3SnBr3 and Br(CH2)4SnBr3 with 3 equiv of but-3-enylmagnesium bromide yielded Br(CH2)3Sn(CH2CH2CH=CH2)3 (3) and Br(CH2)4Sn(CH2CH2CH=CH2)3 (4). Both dendritic branches can be converted into their corresponding Grignard reagents, whose consequent treatment with 0.25 M amounts of SnCl42)3Sn(CH2CH2CH=CH2)3]4 (5) and Sn[(CH2)4Sn(CH2CH2CH=CH2)3]4 (6), respectively. The subsequent hydrostannation of 5 and 6 delivered Sn{(CH2)3Sn[(CH2)4SnPh3]3}4 (7) and Sn{(CH2)4Sn[(CH2)4SnPh3]3}4 (8) as dendrimers of the second generation. All compounds were characterized by elemental analysis, 1H, 13C, and 119Sn NMR spectroscopy, and MALDI-TOF mass spectrometry. resulted in the formation of the dendrimers Sn[(CH
Organometallics, 25 (14), 3370 -3378, 2006
Comment P-31 NMR and p31-pt J-coupling
Platinum(II) Phosphido Complexes as Metalloligands. Structural and Spectroscopic Consequences of Conversion from Terminal to Bridging Coordination
Abstract:
Treatment of the terminal phosphido complexes Pt(dppe)(Me)(PPh(R)) (R = Ph (1), i-Bu (6)) with Pt(dppe)(Me)(OTf) gave the cationic phosphido complexes [(Pt(dppe)(Me))2(PPh(R))][OTf] (R = Ph (7), i-Bu (8)). Similarly, Pt((R,R)-Me-Duphos)(Me)(PPh(i-Bu)) (10) was converted to [(Pt((R,R)-Me-Duphos)(Me)) 2 (PPh(i-Bu))][OTf] (11). A fluxional process in 8 and 11, presumably involving hindered rotation about the Pt-PPh(i-Bu) bonds, was observed by NMR spectroscopy; it resulted in two diastereomers for 8 and four for 11 at low temperature. Coordination of the metalloligand 10 to the [Pt((R,R)-Me-Duphos)(Me)]+11, resulted in structural changes at the Pt-phosphido group, whose geometry changed from distorted pyramidal to tetrahedral. Decomposition of 6 also gave the cation 8, while oxidation of 6 with H2O2 gave the crystallographically characterized phosphido oxide complex Pt(dppe)(Me)(P(O)Ph(i-Bu)) (12).
Saturday, July 29, 2006
Chemistry - A European Journal, Volume: 12, Issue: 20 , pp. 5282 - 5292
Abstract: A number of polycrystalline copper(i) O,O’-dialkyldithiophosphate cluster compounds with Cu4, Cu6, and Cu8 cores were synthesized and characterized by using extended X-ray absorption fine-structure (EXAFS) spectroscopy.The structural relationship of these compounds is discussed. The polyACHTUNGTRENUNGcrystalline copper(i) O,O’-diisobutyl ACHTUNG- TRENUNGdithiophosphate cluster compounds,[Cu8ACHTUNGTRENUN {S2PACHTUNG- TRENUNG(OiBu)2}6(S)]and [Cu6ACHTUNGTRENUNG{S2PACHTUNGTRENUNG(OiBu)2}6], were also characterized by using 31P CP/MAS NMR CP = cross polarization, MAS =magic-angle spinning) and static 65Cu NMR spectroscopies (at different magnetic fields) and powder X-ray diffraction (XRD) analysis. Comparative analyses of the 31P chemical-shifttensor, and the 65Cu chemical shift and quadrupolar-splitting parameters, estimated from the experimental NMR
spectra of the polycrystalline copper(i) cluster compounds, are presented. The adsorption mechanism of the potassium O,O’-diACHTUNGTRENUNGisobutyldithiophosphate collector, KACHTUNGTRENUNG[S2PACHTUNGTRENUNG(OiBu)2], at the surface of synthetic chalcocite (Cu2S) was studied by means of solid-state 31P CP/MAS NMR spectroscopy and scanning electron microscopy (SEM). 31P NMR resonance lines from collector-treated chalcocite surfaces were assigned to a mixture of [Cu8ACHTUNGTRENUNG {S2PACHTUNGTRENUNG (OiBu)2}- 6(S)]and [Cu6- ACHTUNGTRENUNG{S2PACHTUNGTRENUNG(OiBu)2}6]compounds .
Friday, July 28, 2006
J. Chem. Phys. 125, 044510
Ramesh Ramachandran and Robert G. Griffin
Using an analytical model based on multipole-multimode Floquet theory (MMFT), we describe the polarization loss (or depolarization) observed in double-quantum (DQ) dipolar recoupling magic angle spinning (MAS) experiments. Specifically, the factors responsible for depolarization are analyzed in terms of higher order corrections to the spin Hamiltonian in addition to the usual phenomenological decay rate constant. From the MMFT model and the effective Hamiltonians, we elucidate the rationale behind the inclusion of a phenomenological damping term in DQ recoupling experiments. As a test of this theoretical approach, the recoupling efficiency of one class of 13C–13C and 13C–15N resonance width dipolar recoupling experiments are investigated at different magnetic field strengths and compared with the more exact numerical simulations. In contrast to existing analytical treatments, the role of higher order corrections is clearly explained in the context of the MMFT approach leading to a better understanding of the underlying spin physics. Furthermore, the analytical model presented herein provides a general framework for describing coherent and incoherent effects in homonuclear and heteronuclear DQ MAS recoupling experiments.
J. Chem. Phys. 125, 034507
Sergey V. Dvinskikh, Kazutoshi Yamamoto and Ayyalusamy Ramamoorthy
This paper presents a theoretical, numerical, and experimental study of a new class of separated local field (SLF) techniques. These techniques are based on the heteronuclear isotropic mixing leading to spin exchange via the local field (HIMSELF). It is shown that highly efficient and robust SLF experiments can be designed based on double channel windowless homonuclear decoupling sequences. Compared to rotating frame techniques based on Hartmann-Hahn cross polarization, the new approach is less susceptible to the frequency offset and chemical shift interaction and can be applied in the structural studies of macromolecules that are uniformly labeled with isotopes such as 13C and 15N. Furthermore, isotropic mixing sequences allow for transfer of any magnetization component of one nucleus to the corresponding component of its dipolar coupled partner. The performance of HIMSELF is studied by analysis of the average Hamiltonian and numerical simulation and is experimentally demonstrated on a single crystalline sample of a dipeptide and a liquid crystalline sample exhibiting motionally averaged dipolar couplings.
J. Chem. Phys. 124, 204717
T. Shiroka, G. Fumera, O. Ligabue, and M. Riccò G. C. Antonioli
The dynamic and structural properties of the ammonia-doped superconducting fulleride (NH3)xNaK2C60 (0.5x1), well known for its anomalous decrease of transition temperature with doping, have been investigated using sodium and deuterium solid-state NMR techniques. The independence of 23Na quadrupole splitting from the ammonia content x, which, at the same time, substantially affects Tc, suggests a marginal role of the cation position in the superconducting mechanism. On the other hand, a strong reduction of the deuterium quadrupole coupling with respect to the free ammonia value denotes the presence of weak hydrogen bonds between the deuterium atoms and fullerene orbitals. Despite the bond weakness, as evinced by the lively ammonia rotational dynamics even at very low temperatures, the resulting electron localization could explain the observed Tc anomaly. The motion of the ND3–Na group (located in the compound's octahedral voids), as well as the evolution of the ammonia dynamics as a function of temperature, were determined from deuterium NMR line shape analysis and from detailed numerical simulations. While at the lowest measured temperatures only the ammonia rotation around its own C3 axis takes place, above ~25 and 70 K, respectively, also the wobbling of the C3 axis and the ND3 relocation become active, successfully modeled by a strongly correlated motion involving two different time scales
Phys. Chem. Chem. Phys., 2006, 8, 3552 - 3556
Tomer Zidki, Haim Cohen and Dan Meyerstein
Comment: This paper doen not have NMR in it.
Silver and gold nanoparticles are very efficient catalysts for the dimerization of methyl-radicals in aqueous solutions. The rate constants for the reaction of methyl-radicals with the gold and silver nanoparticles were measured and found to be 3.7 × 108 M–1 s–1 and 1.4 × 109 M–1 s–1, respectively. The results thus suggest that alkyl-radicals, also not reducing ones, are scavenged by these nanoparticles. This might explain the role, if such a role exists, of these nanoparticles in medical applications.
Phys. Chem. Chem. Phys., 2006, 8, 3510 - 3519
Christopher I. Ratcliffe, Kui Yu, John A. Ripmeester, Md. Badruz Zaman, Cristina Badarau and Shanti Singh
Solid state 113Cd, 77Se, 13C and 31P NMR have been used to study a number of Cd chalcogenide nanoparticles synthesized in tri-n-octyl-phosphine (TOP) with different compositions and architectures. The pure CdSe and CdTe nanoparticles show a dramatic, size-sensitive broadening of the 113Cd NMR line, which can be explained in terms of a chemical shift distribution arising from multiple Cd environments. From 13C NMR, it has been discovered that TOP, or its derivatives such as TOPO (trioctylphosphine oxide), is rapidly moving about the surface of the nanoparticles, indicating that it is relatively weakly bound as compared to other materials used as surface ligands, such as hexadecylamine. 31P NMR of the nanoparticles shows at least five species arising from coordination of the ligands to different surface sites. 113Cd NMR of CdSeTe alloy and layered nanoparticles has provided crucial information which, in conjunction with results from other techniques (especially optical characterization), has made it possible to develop a detailed picture of the composition and structure of these materials: (i) a true CdSeTe homogeneous alloy nanoparticle, (ii) a nanoparticle segregated into an alloy core region rich in Te, with a CdSeTe (close to 1 : 1 Se : Te) alloy shell and (iii) a CdSe/CdTe/CdSe layered nanoparticle in which the CdTe layer contains a small amount of Se and which forms a Quantum Dot Quantum Well (QDQW) system. The results demonstrate that solid state NMR is a vital tool in the arsenal of characterisation techniques available for nanomaterials.
Phys. Chem. Chem. Phys., 2006, 8, 3423 - 3431
Sharon E. Ashbrook, Laurent Le Pollès, Régis Gautier, Chris J. Pickard and Richard I. Walton
The distorted perovskites NaTaO3 and NaNbO3 have been studied using 23Na multiple-quantum (MQ) MAS NMR. NaTaO3 was prepared by high temperature solid state synthesis and the NMR spectra are consistent with the expected room temperature structure of the material (space group Pbnm), with a single crystallographic sodium site. Two samples of NaNbO3 were studied. The first, a commercially available sample which was annealed at 900 °C, showed two crystallographic sodium sites, as expected for the room temperature structure of the material (space group Pbcm). The second sample, prepared by a low temperature hydrothermal method, showed the presence of four sodium sites, two of which match the expected room temperature structure and the second pair, another polymorph of the material (space group P21ma). This is consistent with powder X-ray diffraction data which showed weak extra peaks which can be accounted for by the presence of this second polymorph. Density functional theory (DFT) calculations support our conclusions, and aid assignment of the NMR spectra. Finally, we discuss the measured NMR parameters in relation to other studies of sodium in high coordination sites in the solid state.
Phys. Chem. Chem. Phys., 2006, 8, 3418 - 3422
Nicolas Mifsud, Bénédicte Elena, Chris J. Pickard, Anne Lesage and Lyndon Emsley
We show how powder samples at natural isotopic abundance can be assigned to crystal structures by using high-resolution proton and carbon-13 solid-state NMR spectra in combination with first principles calculations. Homonuclear proton double-quantum spectra in combination with through-bond proton–carbon HSQC spectra are used to assign the NMR spectra. We then show that the proton chemical shifts can be included in the process of assigning the spectra to a crystal structure using first principles calculations. The method is demonstrated on the K salt of penicillin G.
Phys. Chem. Chem. Phys., 2006, 8, 3379 - 3382
David L. Bryce
Quantum chemical calculations provide new insights into the dependence of J(N,N) coupling tensors on bonding environment in a series of polynitrogen species including N5+.
Phys. Chem. Chem. Phys., 2006, 8, 2733 - 2743
Mathew J. Willans, Bryan A. Demko and Roderick E. Wasylishen
A solid-state nuclear magnetic resonance and zeroth-order regular approximation density functional theory, ZORA-DFT, study of one-bond nuclear spin–spin coupling between group-14 nuclei and quadrupolar 35/37Cl nuclei in triphenyl group-14 chlorides, Ph3XCl (X = C, Si, Ge, Sn and Pb), is presented. This represents the first combined experimental and theoretical systematic study of spin–spin coupling involving spin-pairs containing quadrupolar nuclei. Solid-state NMR spectra have been acquired for all compounds in which X has a spin-1/2 isotope—13C, 29Si, [117/119]Sn and 207Pb—at applied magnetic fields of 4.70, 7.05 and 11.75 T. From simulations of these spectra, values describing the indirect spin–spin coupling tensor—the isotropic indirect spin–spin coupling constant, 1J(X,35/37Cl)iso and the anisotropy of the J tensor, 1J(X,35/37Cl)—have been determined for all but the lead–chlorine spin-pair. To better compare the indirect spin–spin coupling parameters between spin-pairs, 1Jiso and 1J values were converted to their reduced coupling constants, 1Kiso and 1K. From experiment, the sign of 1Kiso was found to be negative while the sign of 1K is positive for all spin-pairs investigated. The magnitude of both 1Kiso and 1K was found to increase as one moves down group-14. Theoretical values of the magnitude and sign of 1Kiso and 1K were obtained from ZORA-DFT calculations and are in agreement with the available experimental data. From the calculations, the Fermi-contact mechanism was determined to provide the largest contribution to 1Kiso for all spin-pairs while spin-dipolar and paramagnetic spin–orbit mechanisms make significant contributions to the anisotropy of K. The inclusion of relativistic effects was found to influence K(Sn,Cl) and K(Pb,Cl).
Phys. Chem. Chem. Phys., 2006, 8, 2635 - 2641
Magnetic alignment of aqueous CTAB in nematic and hexagonal liquid crystalline phases investigated by spin-1 NMR
Jacalyn S. Clawson, Gregory P. Holland and Todd M. Alam
Spin-1 NMR has been used to characterize the magnetically aligned nematic and hexagonal liquid crystalline phases of aqueous cetyltrimethylammonium bromide (CTAB). A nematic/hexagonal biphasic region has been identified for the first time in this system. The nematic phase is characterized by an order parameter of smaller magnitude and greater temperature dependence. Magnetic alignment kinetic rates of the two phases differ greatly, with the nematic phase showing magnetic alignment much faster than the hexagonal phase. Equilibration has been monitored over time by measuring the change in quadrupole splitting as a function of temperature. As the sample equilibrates the temperature dependence of the splitting decreases logarithmically. This work also demonstrates how the phase and order of the liquid crystal can be manipulated during the early part of equilibration.
Friday, July 14, 2006
Joel - Monthly Journal Update
W. Huang et al.
J.Phys.Chem.B(2006)110,12340.
Abstract:
Paramagnetic Eu-substituted Keggin oxopolytungstates crystallize in different forms, determined by the nature of the counterions. The crystal packing is in turn responsible for the variations in the geometry of paramagnetic Eu sites with respect to the anion core. We probed the paramagnetic environments in a series of Eu-substituted Keggin solids, by 31P magic angle spinning NMR spectroscopy. 31P spinning sideband envelopes are dominated by the electron-nuclear dipolar interaction. For the compounds under investigation, both the magnitude and the asymmetry parameter of the electron-nuclear dipolar coupling tensor are sensitive to the mutual arrangements of paramagnetic Eu sites in the crystal lattice. and also report on the stoichiometry of the anion. The electron-nuclear dipolar coupling tensors were calculated from the crystallographic coordinates and the experimentally determined effective magnetic moments, assuming a point dipole approximation. The computed tensors are in very good agreement with the experimental spectra. Furthermore, the P-Eu distance estimates, accurate to within 0.06-0.12 A, can be obtained directly from the magnitude of the electron-nuclear dipolar coupling. This work demonstrates that 31P MAS NMR spectroscopy is a useful probe for investigating local environments in paramagnetic Keggin solids.
Cory
Oxygen Sites and Network Coordination in Sodium Germanate Glasses and Crystals: High Resolution Oxygen-17 and Sodium-23 NMR.
L.S. Du and J.F. Stebbins
J.Phys.Chem.B(2006)110,12427.
Abstract:
Sodium germanate glasses are well-studied materials in which, unlike silicates but analogous to borates, the major structural consequence of alkali addition is generally thought to involve a coordination number increase of the network-forming Ge cations. However, the nature of this change, in particular quantifying fractions of nonbridging oxygens and of five- and/or six-coordinated Ge, has remained unresolved. We present here highresolution 17O results, including triple-quantum MAS NMR (3QMAS), on a series of crystalline model compounds that allow the definition of ranges of chemical shifts corresponding to oxygens bonded to various coordinations of Ge. These include quartz- and rutile-structured GeO2, Na4Ge9O20, Na2Ge4O9, and Na2GeO3 (germanium dioxide, sodium enneagermanate, sodium tetragermanate, and sodium metagermanate). 3QMAS spectra of Na-germanate glasses ranging from 0% to 27% Na2O clearly show the development of partially resolved peaks as alkali is added, corresponding to signals from nonbridging oxygens (in the highest Na glasses) and to oxygen bridging between one four-coordinated and one higher coordinated Ge. As in conventional models of this system, nonbridging oxygen contents are much lower than in corresponding silicates. Although we do not directly distinguish between five- and six-coordinated Ge, modeling of bridging oxygen populations and comparison with measured speciation suggest that substantial proportions of both species are likely to be present. High-field 23Na MAS NMR shows systematic decreases in mean Na-O bond distance and/or coordination number with increasing alkali content that can be compared with published results for high-temperature liquids. These results, as well as comparison of molar volumes of glasses and high-temperature liquids, suggest the possibility of significant temperature effects on liquid structure.
Andy
Photochemical Fine-Tuning of Luminescent Coloout on Cadmium Selenide Nanoparticles: Fabricating a Single-Source Multicolor Luminophore.
T. Torimoto et al.
J.Phys.Chem.B(2006)110,13314.
Abstract:
Size-selective photoetching was applied to silica-coated cadmium selenide (SiO2/CdSe) nanoparticles to precisely control their photoluminescence properties. The absorption spectra of CdSe was blue-shifted by irradiation of monochromatic light, and finally, the absorption onset agreed with the wavelength of irradiation light, indicating that CdSe particles were photoetched to smaller ones until the irradiated photons were not absorbed by the photoetched particles and that the SiO2 shell layer surrounding the CdSe core prevented coalescence between the photoetched particles. Although as-prepared SiO2/CdSe did not exhibit photoluminescence, the application of size-selective photoetching to SiO2/CdSe resulted in the development of the band gap emission, with the degree being enhanced with progress of the photoetching. The peak wavelength of photoluminescence decreased with a decrease in the wavelength used for the photoetching, so that the luminescence color could be tuned between red and blue. Partial photoetching of SiO2/CdSe nanoparticle films produced intense band gap emission of CdSe at the photoetched area, while the remainder of the SiO2/ CdSe films did not exhibit detectable photoluminescence, resulting in the formation of a clear photoluminescence image under UV irradiation. This technique makes it possible to produce a multicolored photoluminescence image by irradiation with monochromatic lights having various wavelengths using a single source material.
Josh, Andy
Synthesis, crystal structure and magnetic properties of an alternating manganese chain.
M.R. Silva et al.
JSolidStateChem (2006) 179, 2054.
Abstract:
A new 1D complex has been prepared and characterized. X-ray single crystal structure con.rms that the Mn(II) ions assemble in alternating chains with Mn–Mn distances of 3.8432(13) and 4.4428(14)A . A 3D network of hydrogen bonds links the chains together. The temperature dependence of the magnetic susceptibility reveals that this compound undergoes a magnetic transition and exhibits an antiferromagnetic interaction in the low-temperature phase with two alternating exchange interactions of -2.32(1) and -5.55(1)cm-1.
Rob
Sr4PbPt4O11, the first platinum oxide containing Pt2 6+ ions.
C. Renard et al.
JSolidStateChem (2006) 179, 2054.
Abstract:
We report the synthesis and crystal structure of the new compound Sr4PbPt4O11, containing platinum in highly unusual square pyramidal coordination. The crystals were obtained in molten lead oxide. The structure was solved by X-ray single crystal diffraction techniques on a twinned sample, the final R factors are R = 0.0260 and wR = 0.0262. The symmetry is triclinic, space group P1¯ , with a = 5.6705(6) A, b = 9.9852(5) A, c = 10.0889(5) A, alpha = 90.421(3), beta = 89.773(8), gamma = 90.140(9) and Z = 2. The structure is built from dumbell-shaped Pt2O9 entities formed by a dinuclear metal–metal bonded Pt2 6+ ion with asymmetric environments of the two Pt atoms, classical PtO4 square plane and unusual PtO5 square pyramid. Successive Pt2O9 entities deduced from 901 rotations are connected through the oxygens of the PtO4 basal squares to form (Pt4O10)-8 columns further connected through Pb2+ and Sr2+ ions. Raman spectroscopy confirmed the peculiar platinum coordination environment.
Structural and 31P NMR investigation of Bi(MM')2PO6 statistic solid solutions: Deconvolution of lattice constrants and cationic influences.
M. Colmont et al.
JSolidStateChem (2006) 179, 2111.
Abstract:
Two solid solutions BiMxMg(2-x)PO6 (with M2+ = Zn or Cd) have been studied through 31P MAS NMR. The analysis has been performed on the basis of re.ned crystal structures through X-ray diffraction and neutron diffraction. The BiZnxMg(2-x)PO6 does not provide direct evidence for sensitive changes in the phosphorus local symmetry. This result is in good agreement with structural data which show nearly unchanged lattices and atomic separations through the Zn2+ for Mg2+ substitution. On the other hand, the Cd2+ for Mg2+ substitution behaves differently. Indeed, up to five resonances are observed, each corresponding to one of the five first-cationic neighbour distributions, i.e. 4Mg/0Cd, 3Mg/1Cd, 2Mg/2Cd, 1Mg/3Cd and 0Mg/4Cd. Their intensities match rather well the expected weight for each con.guration of the statistical Cd2+/Mg2+ mixed occupancy. The match is further improved when one takes into account the in.uence of the 2nd cationic sphere that is available from high-.eld NMR data (18.8 T). Finally, the fine examination of the chemical shift for each resonance versus x allows to de-convolute the mean Z/a2 effective field into two sub-effects: a lattice constraint only term and a chemical-only term whose effects are directly quantifiable.
JACS - Volume 128, Issue 27
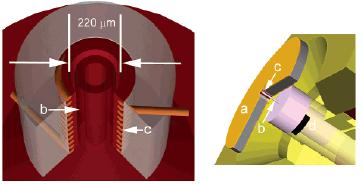
Cory's Comment: small coils = super high (600 kHz) proton decoupling fields!
Title: Microcoil High-Resolution Magic Angle Spinning NMR Spectroscopy
Authors: Hans Janssen, Andreas Brinkmann, Ernst R. H. van Eck, P. Jan M. van Bentum, and Arno P. M. Kentgens
Page #: 8722
Abstract: We report the construction of a dual-channel microcoil nuclear magnetic resonance probehead allowing magic-angle spinning for mass-limited samples. With coils down to 235 m inner diameter, this allows high-resolution solid-state NMR spectra to be obtained for amounts of materials of a few nanoliters. This is demonstrated by the carbon-13 spectrum of a tripeptide and a single silk rod, prepared from the silk gland of the Bombyx mori silkworm. Furthermore, the microcoil allows for radio frequency field strengths well beyond current probe technology, aiding in getting the highest possible resolution by efficiently decoupling the observed nuclei from the abundantly present proton nuclei.
Thursday, July 13, 2006
Cory's Journals - June 2006
As per usual, by the end of today, I will have the journal articles that I collected in June 2006 available in their usual spot.
Peace.
