Monday, January 31, 2011
J. Phys. Chem. B, 2011, 115 (4), pp 730–736
Thiamin diphosphate (ThDP) is a key coenzyme in sugar metabolism. The 4′-aminopyrimidine ring of ThDP cycles through several ionization and tautomeric states during enzyme catalysis, but it is not fully understood which states are adopted during the individual steps of the catalytic cycle. Thiamin has been synthesized with labels selectively inserted into the C2 and C6′ positions, as well as into the amino group, creating [C2, C6′-13C2] thiamin and [N4′-15N] thiamin. Magic-angle spinning (MAS) NMR spectroscopy has been employed to record the 13C and 15N chemical shift anisotropy (CSA) tensors for C2, C6′, and N4′ atoms. Our results indicate that the isotropic chemical shifts as well as the principal components of the 13C and 15N CSA tensors are very sensitive to the protonation states in these compounds and, therefore, permit differentiating between the two ionization states, 4-aminopyrimidine and 4-aminopyrimidinium. Using density functional theory (DFT), we have calculated the magnetic shielding anisotropy tensors of C2, C6′, and N4′ and found excellent agreement between the computed and the experimental tensors. Our findings indicate that MAS NMR spectroscopy in conjunction with DFT calculations is a sensitive probe of ionization states in the thiamin cofactor. The results of this study will serve as a guide for characterization of ionization and tautomeric states of thiamin in complexes with thiamin-dependent enzymes.
Siting and Mobility of Deuterium Absorbed in Cosputtered Mg0.65Ti0.35. A MAS 2H NMR Study
J. Phys. Chem. C, 2011, 115 (1), pp 288–297
Nanostructured magnesium titanium alloys are interesting lightweight materials for chemical hydrogen storage. We have therefore investigated the siting and dynamics of deuterium absorbed in a Mg0.65Ti0.35 alloy generated by magnetron cosputtering, and made a comparison to the corresponding features in bulk samples of deuterium-loaded Mg0.65Ti0.35 and Mg0.65Sc0.35 prepared by ball-milling and melt-casting, respectively. Magic-angle spinning 2H NMR of cosputtered Mg0.65Ti0.35D1.1 shows partly resolved signals of deuterium located in nonconductive domains at tetrahedral Mg4 and mixed MgnTi4−n sites (4 ppm) and deuterium at Ti4 sites in conducting TiD2 nanodomains (−29 and −68 ppm). No bulk TiD2 signal at −150 ppm is observed, in contrast to what we find in ball-milled Mg0.65Ti0.35D0.65, which is largely phase separated. The deuterium species with shift values of 4 and −29 ppm undergo complete exchange at a subsecond time scale in one- and two-dimensional exchange NMR and must therefore be close together in the lattice. In contrast, deuterium resonating at −68 ppm does not show deuterium exchange and thus appears to be located at more stable sites. The observed deuterium exchange and the reduced Knight shift compared to bulk TiD2 are explained using a model with TiD2 nanoslabs.
Towards Portable High-Resolution NMR Spectroscopy†
Angew. Chem. Int. Ed. 2011, 50, 354 – 356
Analysis on the go: Portable high-resolution NMR spectroscopy is of great interest for many applications. Recent advances in magnet design, spectrometer stability, and acquisition schemes have placed the realization of low-field spectrometers based on room-temperature permanent magnets and that can deliver chemical shift resolution within reach.
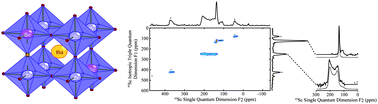
Probing the local structures and protonic conduction pathways in scandium substituted BaZrO3 by multinuclear solid-state NMR spectroscopy
from RSC - J. Mater. Chem. latest articles by Clare P. GreyA comprehensive multinuclear solid-state NMR study of scandium-substituted BaZrO3 is reported. Static low field and MQMAS very high field 45Sc NMR data revealed the presence of both 5- and 6-coordinated scandium atoms, 5-coordinated scandium arising from Sc nearby an oxygen vacancy. 17O NMR spectra showed the presence of up to three different chemical oxygen environments assigned to Zr–O–Zr, Zr–O–Sc and Sc–O–Sc. From the ratios of these different oxygen sites, the distribution of the scandium cations was close to random but indicated that the maximum scandium incorporation was lower than expected, consistent with the observation of Sc2O3 impurities at substitution levels of 30% Sc for Zr. 1H and 45Sc NMR data on the hydrated materials revealed the presence of scandium next to protonic defects. Finally, variable temperature 1H NMR showed the presence of at least two different proton environments in between which proton transfer occurs at ambient temperatures (300 K).
Solid state NMR study on the thermal decomposition pathway of sodium amidoborane NaNH2BH3
from RSC - J. Mater. Chem. latest articles by Yoshitsugu KojimaThe thermal decomposition pathway of sodium amidoborane (NaAB; NaNH2BH3) has been investigated in detail by using solid state NMR spectroscopy. 23Na MAS/3QMAS NMR spectra suggested that NaH and an amorphous Na–N–B–H phase started to be formed as decomposition products even at 79 °C, although NaAB was prepared from NaH and NH3BH3 by ball milling at room temperature. Based on the quantitative analyses of the 23Na MAS spectra, we proposed a decomposition reaction to 200 °C to be NaNH2BH3 → Na0.5NBH0.5 + 0.5NaH + 2.0H2. The hypothetical phase Na0.5NBH0.5 is amorphous, where the basic molecular unit of the original NaAB is polymerized into a [–B![[double bond, length as m-dash]](http://www.rsc.org/images/entities/char_e001.gif) N–]n network structure. It was also found that the diammoniate of diborane (DADB) and polyaminoborane (PAB) were not formed during the decomposition of NaAB, which are both key compounds on the pyrolysis of ammonia borane (AB).
N–]n network structure. It was also found that the diammoniate of diborane (DADB) and polyaminoborane (PAB) were not formed during the decomposition of NaAB, which are both key compounds on the pyrolysis of ammonia borane (AB).
Monday, January 17, 2011
17O Central Transition NMR
Quadrupole Central Transition 17O NMR Spectroscopy of Biological Macromolecules in Aqueous Solution
Jianfeng Zhu and Gang Wu*
J. Am. Chem. Soc., Article ASAP
DOI: 10.1021/ja1079207
Abstract: We demonstrate a general nuclear magnetic resonance (NMR) spectroscopic approach in obtaining high-resolution 17O (spin-5/2) NMR spectra for biological macromolecules in aqueous solution. This approach, termed quadrupole central transition (QCT) NMR, is based on the multiexponential relaxation properties of half-integer quadrupolar nuclei in molecules undergoing slow isotropic tumbling motion. Under such a circumstance, Redfield’s relaxation theory predicts that the central transition, mI = +1/2 ↔ −1/2, can exhibit relatively long transverse relaxation time constants, thus giving rise to relatively narrow spectral lines. Using three robust protein−ligand complexes of size ranging from 65 to 240 kDa, we have obtained 17O QCT NMR spectra with unprecedented resolution, allowing the chemical environment around the targeted oxygen atoms to be directly probed for the first time. The new QCT approach increases the size limit of molecular systems previously attainable by solution 17O NMR by nearly 3 orders of magnitude (1000-fold). We have also shown that, when both quadrupole and shielding anisotropy interactions are operative, 17O QCT NMR spectra display an analogous transverse relaxation optimized spectroscopy type behavior in that the condition for optimal resolution depends on the applied magnetic field. We conclude that, with the currently available moderate and ultrahigh magnetic fields (14 T and higher), this 17O QCT NMR approach is applicable to a wide variety of biological macromolecules. The new 17O NMR parameters so obtained for biological molecules are complementary to those obtained from 1H, 13C, and 15N NMR studies.
Solid-State 17O NMR Spectroscopy of Large Protein–Ligand Complexes†
Dr. Jianfeng Zhu1, Dr. Eric Ye2, Dr. Victor Terskikh3, Prof. Dr. Gang Wu1
Article first published online: 29 OCT 2010
DOI: 10.1002/anie.201002041
Angewandte Chemie International Edition
Volume 49, Issue 45, pages 8399–8402, November 2, 2010
Keywords:
oxygen-17;protein–ligand interactions;proteins;solid-state NMR spectroscopy;structure refinement
Oxygen, oxygen, everywhere! Poor sensitivity has hindered the development of solid-state 17O NMR spectroscopy as a practical technique for the structural elucidation of protein complexes. However, this has now changed and it has been demonstrated that multinuclear 17O, 27Al, 13C NMR parameters can be used to aid structural refinement for a protein-bound ligand molecule (see picture).
Monday, January 10, 2011
Solid-State NMR
A practical guide for the setup of a 1H-31P-13C double cross-polarization (DCP) experiment
Source: Solid State Nuclear Magnetic Resonance, In Press, Accepted Manuscript, Available online 13 December 2010
Wlodzimierz, Ciesielski , Hassan, Kassasir , Marek J., Potrzebowski
Multinuclear NMR study of silica fiberglass modified with zirconia
Source: Solid State Nuclear Magnetic Resonance, In Press, Accepted Manuscript, Available online 29 December 2010
O.B., Lapina , D.F, Khabibulin , V.V., Terskikh
Kinetics of 1H→13C NMR cross-polarization in polymorphs and solvates of the antipsychotic drug olanzapine
Source: Solid State Nuclear Magnetic Resonance, In Press, Accepted Manuscript, Available online 4 January 2011
Waclaw, Kolodziejski , Joanna, Herold , Marzena, Kuras , Irena, Wawrzycka-Gorczyca , Anna E., Koziol
Inorg. Chem.
Solvent Effects and Dynamic Averaging of 195Pt NMR Shielding in Cisplatin Derivatives
Lionel A. Truflandier, Kiplangat Sutter, and Jochen Autschbach*Inorg. Chem., Article ASAP
The influences of solvent effects and dynamic averaging on the 195Pt NMR shielding and chemical shifts of cisplatin and three cisplatin derivatives in aqueous solution were computed using explicit and implicit solvation models. Within the density functional theory framework, these simulations were carried out by combining ab initio molecular dynamics (aiMD) simulations for the phase space sampling with all-electron relativistic NMR shielding tensor calculations using the zeroth-order regular approximation. Structural analyses support the presence of a solvent-assisted “inverse” or “anionic” hydration previously observed in similar square-planar transition-metal complexes. Comparisons with computationally less demanding implicit solvent models show that error cancellation is ubiquitous when dealing with liquid-state NMR simulations. After aiMD averaging, the calculated chemical shifts for the four complexes are in good agreement with experiment, with relative deviations between theory and experiment of about 5% on average (1% of the PtII chemical shift range).
J. Phys. Chem. A
Crystal Structure Based Design of Signal Enhancement Schemes for Solid-State NMR of Insensitive Half-Integer Quadrupolar Nuclei
Luke A. O’Dell* and Christopher I. Ratcliffeluke.odell@nrc-cnrc.gc.ca
A combination of density functional and optimal control theory has been used to generate amplitude- and phase-modulated excitation pulses tailored specifically for the 33S nuclei in taurine, based on one of several reported crystal structures. The pulses resulted in significant signal enhancement (stemming from population transfer from the satellite transitions) without the need for any experimental optimization. This allowed an accurate determination of the 33S NMR interaction parameters at natural abundance and at a moderate magnetic field strength (11.7 T). The 33S NMR parameters, along with those measured from 14N using frequency-swept pulses, were then used to assess the accuracy of various proposed crystal structures.
___________________________________________________
Interactions of Volatile Organic Compounds with Syndiotactic Polystyrene Crystalline Nanocavities
Alexandra R. Albunia*, Patrizia Oliva, and Alfonso Grassi
aalbunia@unisa.it
The interaction of some volatile organic compounds, namely, 1,2-dichloroethane, 1,2-dibromoethane, and 1,1,2,2-tetrachloroethane, included in the δ crystalline phase of syndiotactic polystyrene (sPS) has been studied in terms of conformation, orientation, and dynamical behavior. By combination of X-ray diffraction (XRD), Fourier-transform infrared (FTIR), and solid-state 2H NMR analyses, it has been shown that despite the differences in guest molecular properties (mass, boiling temperature, and volume), stable sPS/guest δ-clathrate cocrystals are formed since the nanoporous δ crystalline form has a flexible structure able to adapt itself to the guest molecule. As a consequence of inclusion, it has been shown that the guest diffusivity is strongly reduced and the dynamical processes are constrained, particularly when these guests are in trans
conformation. This suggests the nanoporous sPS δ form to be an efficient tool for water and air purification through volatile organic compound absorption.
